Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma). One type of atom can form a pure gas; another type of atom can form an elemental molecule (for example, oxygen); and the third type of atom can form a compound molecule (e.g. carbon dioxide).
There are several pure gasses in a gas mixture, such as air. Gasses are distinguished from other substances by their particle-to-particle distance.
When separated using this method, colorless gas is usually invisible to the human eye. We’ll look at the heaviest gasses the universe has in this article. Continue reading to learn more:
1. Radon

Radon is a chemical element found in the atmosphere with the symbol Rn and the atomic number 86. It is a colorless, odorless, and tasteless radioactive noble gas.
It naturally occurs in trace amounts as an intermediate step in the normal radioactive decay chains, where thorium and uranium slowly decay into lead and a variety of other short-lived radioactive elements.
Thorium and uranium decay into lead and a variety of other radioactive elements with short half-lives. Radon is a radioactive element that is the immediate decay product of radium.
Its most stable isotope, 222Rn, has a half-life of only 3.8 days, making it one of the elements on the periodic table with the shortest half-life.
Radon is one of the heaviest gasses known to exist in the universe and is only marginally soluble in water. This element has an atomic weight of 222 atomic mass units. As a result, radon is 220 times heavier than hydrogen.
2. Nitrogen

Nitrogen is a chemical element with the atomic number 7 and the symbol N in its chemical formula. In 1772, a Scottish physician named Daniel Rutherford was the first to discover and isolate the substance.
Even though Carl Wilhelm Scheele and Henry Cavendish accomplished the same thing independently around the same time, Rutherford is generally credited with the achievement because his work was published first.
In 1790, French chemist Jean-Antoine-Claude Chaptal discovered nitrogen in nitric acid and nitrates and proposed the name nitrogène to describe the element.
Nitrogen has an atomic weight of 14.0067, making nitrogen one of the heaviest and most abundant gasses in the universe.
3. Argon

Argon is a naturally occurring chemical element with the symbol ‘Ar’ and the atomic number 18. It belongs to the periodic table’s group 18 and is classified as a noble gas.
Argon is the third most abundant gas in the Earth’s atmosphere, with a concentration of 0.934 percent (9340 ppmv). I
It is more than twice as abundant as water vapor (which averages around 4000 parts per million by volume but varies greatly), 23 times more abundant than carbon dioxide (400 parts per million by volume), and 500 times more abundant than neon (18 ppmv).
Argon is the most abundant noble gas in the Earth’s crust, accounting for 0.00015 percent of the total crustal mass. It is also one of the heaviest gasses known to man, with an atomic weight of 39.948 u.
4. SF5C(O)CF3
Source: Wikipedia
This is probably the heaviest sulfur-containing gas known to science, with a molecular weight of 224 and a boiling point of 15.6 degrees Celsius – 1.5 times heavier than the well-known SF6 and 7.8 times heavier than air.
5. C4F10 and another C4F10
Source: Wikipedia
Despite having the same molecular weight (238), their structural differences are significant. The first is perfluorobutane, and the second is perfluorobutane, with boiling temperatures of -1.7 and 0 degrees Celsius, respectively.
Because it is used for “microbubble ultrasound contrast agents,” the first one may be less toxic. Under normal conditions, sound can travel at only 119 meters per second in that gas, which is enough to transform the fifth octave C into the third octave F#.
6. Tellurium Hexafluoride (TeF6)

According to the manufacturer, “it is a colorless, highly toxic gas with an extremely unpleasant odor.” It is one of the heaviest gasses known to exist in the universe due to its extremely high molecular mass of 241.59 g/mol.
7. Pentafluoro Lodoethane (CF3CF2I )
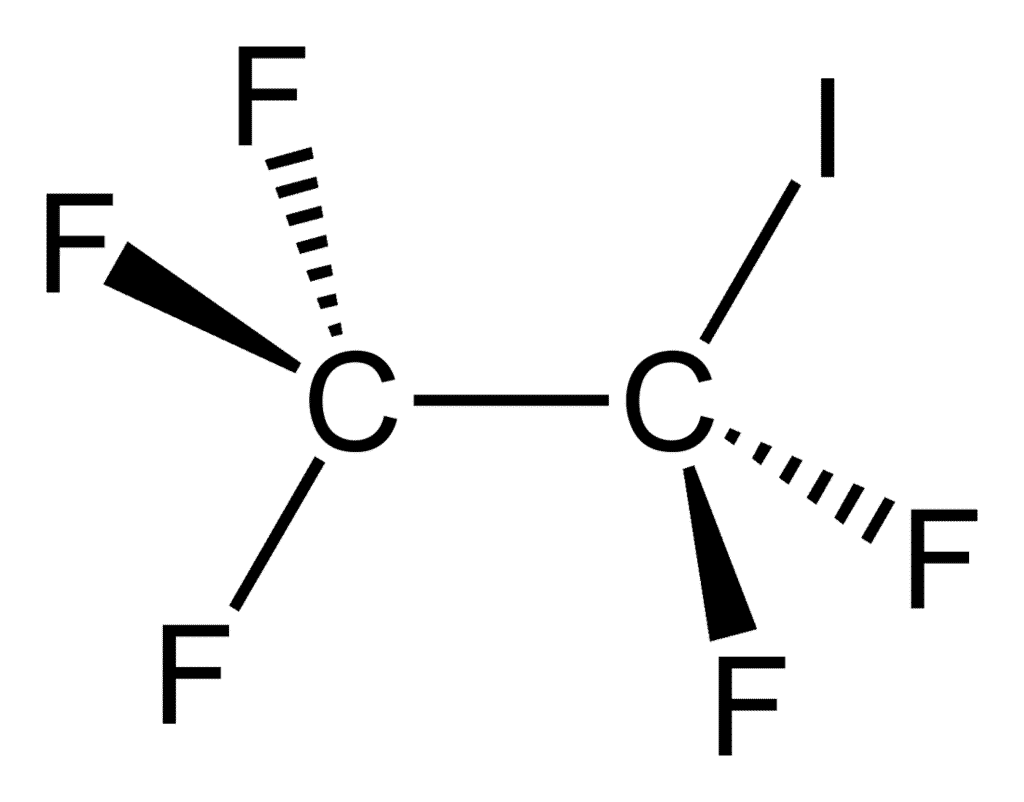
To make a new compound, take a standard ethane C2H6, replace one hydrogen with iodine, and the rest with fluorine.
Although it is listed as a potential anesthetic, it is not poisonous – at least not if knocking oneself out or overdosing is excluded. It is one of the heaviest gasses on the planet, with a molar mass of 353.824 g/mol.
8. Tungsten Hexafluoride ( WF6)

With caution, Wikipedia describes it as “one of the heaviest gasses,” but does not provide any examples of anything even heavier. WF6 is a chemical that has been extensively researched and is used in the semiconductor industry to deposit thin layers of Tungsten on Silicon.
Buying this gas to keep it at home, on the other hand, is a bad idea. Because it is corrosive, poisonous, and chemically active, it is more visually appealing as a painting than as an ampule.
WF6 ranks first on the list of the heaviest gasses in the universe due to its high molecular weight of 297.83 g/mol.
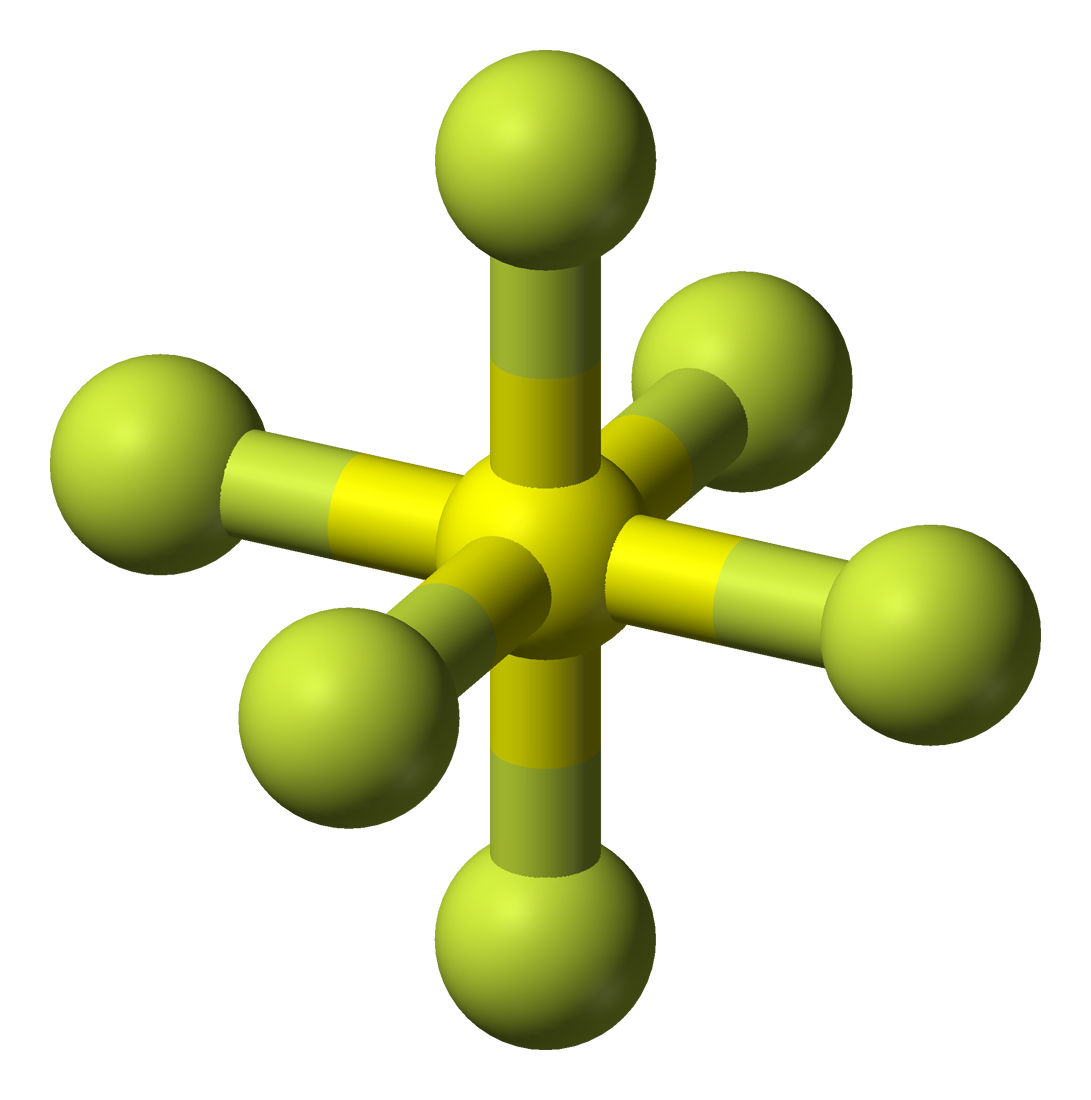
9. Iodine Heptafluoride (IF7)
It is a gas that is nine times heavier than air in density. Chemically reactive and potentially hazardous. “When it comes into contact with organic material, it can cause a fire.” With a molecular weight of 259.9 g/mol, it makes it onto the list of the heaviest gasses.