The building blocks or compounds that makeup peptides and proteins are amino acids.
The amino acid molecule consists of an amino group and a carboxyl group bound to a tetrahedral carbon. This carbon is known as the α-carbon (alpha-carbon).
Aside from their side chains, amino acids can also be distinguished by their R groups. There will be differences in the structure, electrical charge, and polarity of each amino acid’s R group.
If you’re interested in a list of the heaviest to lightest amino acids, stay tuned to this piece to learn more:
1. Tryptophan
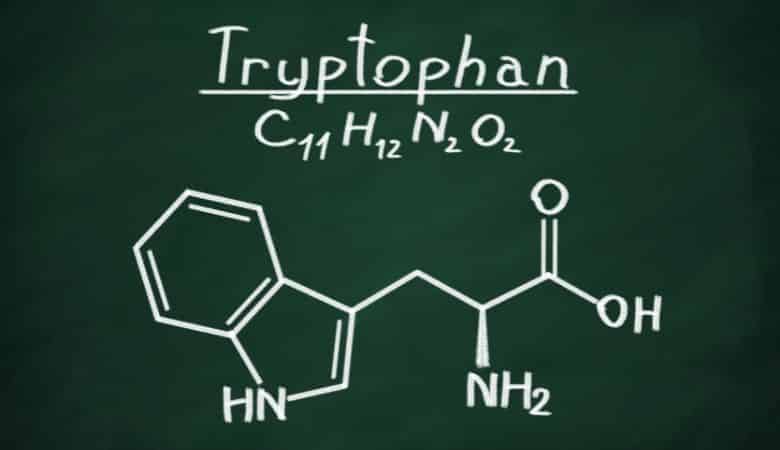
Tryptophan (symbol Trp or W) is an α-amino acid required for protein synthesis. Specifically, tryptophan contains three amino acid groups: an amino group, a carboxylic acid group, and a side chain indole, making it a nonpolar aromatic amino acid. As a result, it cannot be synthesized by the body and must be obtained from food.
Additionally, tryptophan is a precursor to serotonin, melatonin, and vitamin B3. It is encoded by the UGG codon. It tops the list of heaviest amino acids with a molecular weight of 204.23.
2. Tyrosine
The amino acid L-Tyrosine or tyrosine (symbol Y or Tyr) is one of the 20 amino acids used in protein synthesis by cells. The amino acid has polar side groups, making it non-essential.
Tyrosine is named after the Greek word for cheese, tyro, as it was discovered in cheese casein in 1846 by German chemist Justus von Liebig.
In the context of a functional group or side chain, it is referred to as tyrosyl. Despite its hydrophobic classification, tyrosine is more hydrophilic than phenylalanine.
A messenger RNA contains the codons UAA and UAU that encode this protein. Tyrosine is one of the heaviest amino acids with a molecular weight of 181.19.
3. Arginine
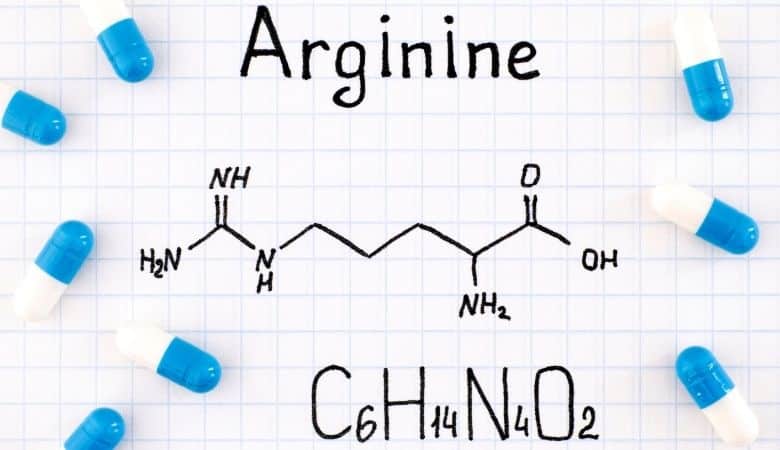
The amino acid arginine, also known as l-arginine (symbol Arg or R), is used by the body to synthesize proteins. This compound consists of an amino group, a carboxylic acid group, and a side chain that is an aliphatic straight chain with three carbons ending in guanidines.
Arginine is a charged, aliphatic amino acid after protonation of the carboxylic acid (*COO*), protonation of the amino group (*NH3+), and protonation of the guanidino group (-C-(NH2)2+). It is the precursor for nitric oxide biosynthesis. This gene is encoded by the codons CGU, CGC, CGA, CGG, AGA, and AGG.
Semi essential and conditionally essential amino acids can be derived from arginine, depending on an individual’s developmental stage and health status. It also passes as one of the heaviest amino acids with a molecular weight of 174.20.
4. Histidine
The amino acid histidine (symbol His or H) is essential for protein synthesis.
It is composed of three amino groups (which are protonated in physiological conditions -NH3+ and carboxylic acid groups that are deprotonated under physiological conditions -COO*) and one imidazole side chain (which is partially protonated), making it a positively charged amino acid under physiological conditions.
In recent studies, it has been shown to be essential to adults as well, despite initially being thought to be essential only in infants.
Codons CAU and CAC are responsible for its encoding. Histidine is one of the heaviest amino acids with a molecular weight of 155.16.
5. Serine
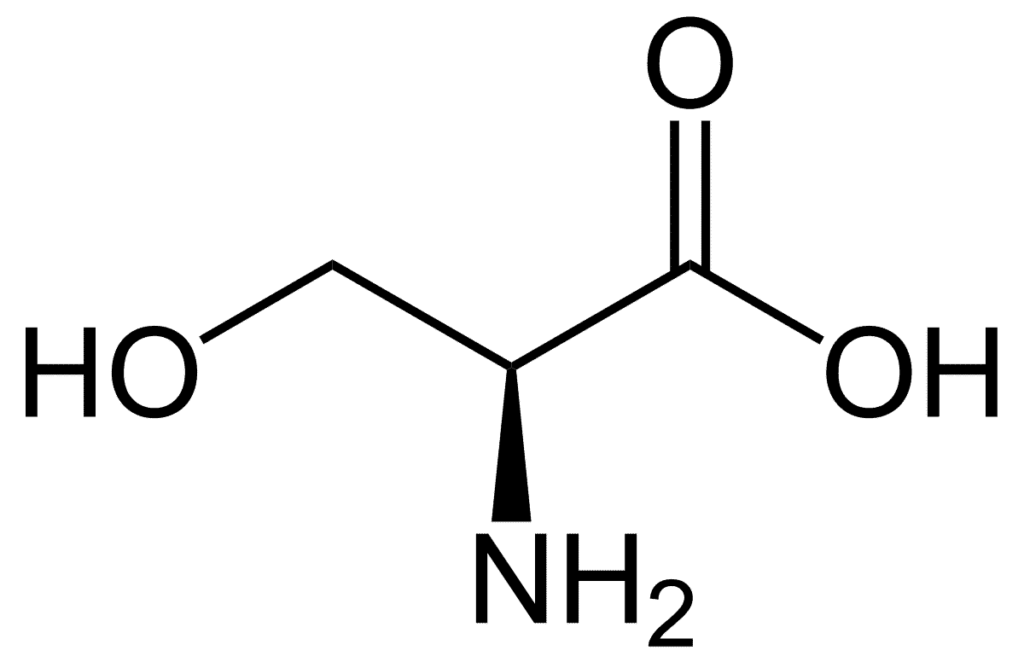
In the biosynthesis of proteins, serine (symbol Ser or S) is an essential amino acid. It consists of an amino group (which is protonated in the biological environment), a carboxyl group (which is deprotonated under biological conditions), and a side chain made up of a hydroxymethyl group, hence its classification as a polar amino acid.
Since it can be synthesized in the human body under normal physiological conditions, it is not essential. There are six codons encoding it: UCU, UCC, UCA, UCG, AGU, and AGC. Serine is one of the lightest amino acids with a molecular weight of 105.9.
6. Alanine
The amino acid alanine (symbol Ala or A) participates in the production of proteins. The central carbon atom carries both an amine group and a carboxylic acid group attached to its side chain, which is a methyl group.
In consequence, its IUPAC systematic name is 2-aminopropanoic acid, and it is classified as an aliphatic, nonpolar α-amino acid. In biological conditions, these molecules exist in their zwitterionic form, in which the amine group protonates (as *NH3+) and the carboxyl group deprotonates (as −CO2−).
It is not essential to humans as it can be synthesized metabolically and does not need to be consumed. GC codons (GCU, GCC, GCA, and GCG) encode it. Alanine is one of the lightest amino acids with a molecular weight of 89.10.
7. Glycine
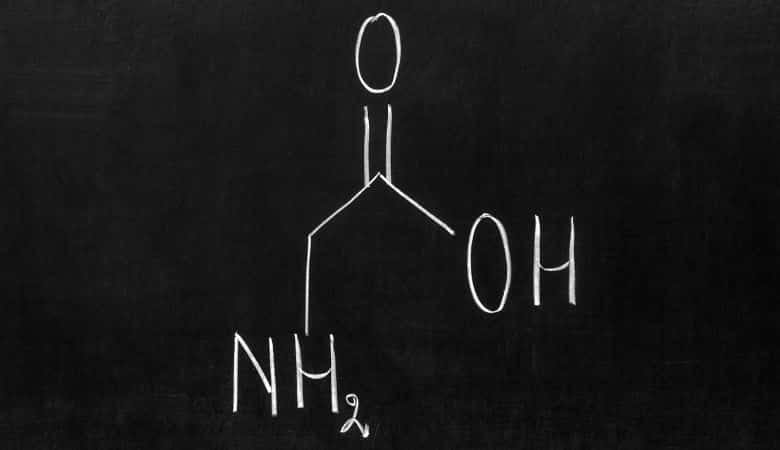
The amino acid glycine (symbol Gly or G) has one hydrogen atom as its side chain. It has the chemical formula NH2CH2COOH, which is the simplest amino acid (carbamic acid is unstable).
Glycine is a proteinogenic amino acid. All the codons that begin with GG encode it (GGU, GGC, GGA, GGG). Due to its compact form, glycine is essential for the formation of alpha-helices in secondary protein structures.
Due to its abundance, it also makes up most of the collagen triple helices. Glucine is also an inhibitory neurotransmitter, so blocking its release within the spinal cord (such as during an infection with Clostridium tetani) can lead to spastic paralysis caused by uninhibited muscle contractions.
Glycine is the lightest amino acid in existence with a molecular weight of 75.07.






